Alysis bufferis usually a buffer option utilized for the objective of bursting open cells for use in molecular biology experiments that evaluate the labile macromolecules of the tissue (elizabeth.g. western blot for protein, or for DNA extraction). Many lysis buffers contain buffering salts (e.g. Tris-HCl) and ionic salts (e.gary the gadget guy. NaCl) to manage the pH and osmolarity of the lysate. Sometimes detergents (such as Triton Times-100 or SDS) are usually added to break up membrane layer constructions. For lysis buffers focused at protein extraction, protease inhibitors are often integrated, and in tough situations may be almost needed. Lysis buffers can become utilized on both animal and seed tissue tissues.1
DNA 'snot' in cell lysis/protein extraction - (Oct/06/2008 ) There is a sticky gooey snot-like substance in my cell lysate that I am told is DNA. It does not dissolve when boiled in SDS, It won't spin down and I don't know how to get rid of it. The lysis buffer (aka solution 2) contains sodium hydroxide (NaOH) and the detergent Sodium Dodecyl (lauryl) Sulfate (SDS). SDS is there to solubilize the cell membrane. NaOH helps to break down the cell wall, but more importantly it disrupts the hydrogen bonding between the DNA bases, converting the double-stranded DNA (dsDNA) in the cell.
- 2Components
- 2.2Additives
- 3Commonly utilized buffers
Selecting a bufferedit
The primary objective of lysis buffer will be isolating the substances of interest and keeping them in a stable environment. For protein, for some experiments, the target protein should be totally denatured, while in some other experiments the focus on proteins should stay folded and functional. Different protein also have got different attributes and are found in different cellular conditions. Therefore, it can be essential to choose the greatest buffer based on the purpose and style of the tests. The essential aspects to end up being considered are usually: pH, ionic strength, utilization of cleaning agent, protease inhibitors to avoid proteolytic procedures.2For illustration, detergent addition is essential when lysing Gram-negative bacteria, but not for Gram-positive bacteria.3It is certainly typical that a protease inhibitor is certainly added to lysis buffer, along with various other enzyme inhibitors of selection, like as a phosphatase inhibitor when learning proteins with phosphorylation.
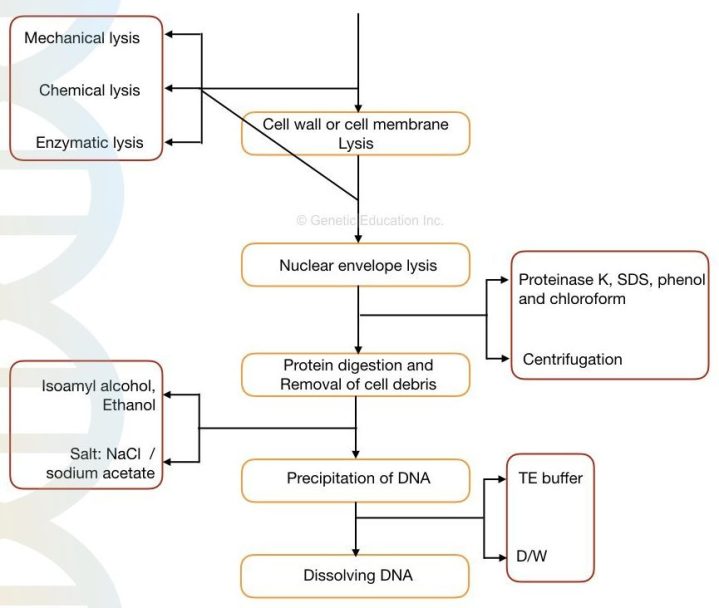
Partsedit
Barrieredit
Buffer creates an environment for separated protein. Each buffer option has a specific pH variety, so the buffer should become chosen based on whether your focus on protein is certainly steady under a certain pH. Furthermore, for buffers with identical pH ranges, it is important to think about whether the buffer can be suitable with your target proteins.4The desk below includes several almost all commonly utilized buffers and their pH ranges.4
| Barrier | pH Range |
|---|---|
| Salt dihydrogen phosphate / disodium hydrogen phosphate | 5.8 - 8.0 |
| Tris - HCl | 7.0 - 9.0 |
| HEPES - NaOH | 7.2 - 8.2 |
Additivesedit
Saltsedit
Lysis buffer usually consists of one or more salts. The functionality of salts in lysis buffer can be to set up an ionic strength in the buffer remedy. Some of the nearly all commonly utilized salts are NaCl, KCl, and (NH4)2Thus4.They are usually used with a focus between 50 and 150 mM.4
Salt Dodecyl Sulfate (SDS) construction
Soapedit
Triton X-100 framework
Detergents are usually natural amphipathic (with hydrophobic end and a hydrophilic head) surfactants. They are usually utilized to distinct membrane proteins from membrane because the hydrophobic component of soap can encompass biological walls and therefore isolate membrane protein from walls.5Although detergents are usually widely used and have got similar functions, it is certainly essential to recognize the actual and chemical properties of the detergents of attention in purchase to determine the optimum one to use for your experiment.
Detergents are usually often categorized as nonionic, anionic, cationic, or zwitterionic, based on their hydrophilic head group function.5
Nonionic detergents like Triton X-100 and zwitterionic detergents like CHAPS (3-(3-cholamidopropyl)dimethylammonio-1-propanesulfonate) are nondenaturing (will not really disrupt proteins functions). Ionic detergents like salt dodecyl sulfate (SDS) and cationic detergents like ethyl trimethyl ammonium bromide are usually denaturing (will disturb protein features).6Detergents are a main component that establishes the lysis power of a provided lysis buffer.
Othersedit
Various other additives include metallic ions, sugar like blood sugar, glycerol, steel chelators (age.gary the gadget guy. EDTA), and decreasing agencies like dithiothreitol (DTT).4
Typically used buffersedit
NP-40 lysis bufferedit
It may become the nearly all widely utilized lysis buffer. The solubilizing agent will be NP-40, which can end up being replaced by additional detergents at various concentrations. Since NP-40 can be a nonionic soap, this lysis buffer offers a milder impact than RIPA buffer. It can be utilized when proteins functions are usually to become retained with minimal interruption.7
Formula:7
- 150 mM NaCl
- 1.0% Nonidet G-40 or Triton X-100
- 50 mM Tris-Cl
- Adjust pH to 7.4
RIPA (RadioImmunoPrecipitation Assay) lysis bufferedit
RIPA buffer will be a typically used lysis buffer for immunoprecipitation and general protein extraction from cells and cells. The buffer can become saved without vanadate at 4 °C for up to 1 yr.8RIPA buffer releases proteins from tissue as properly as disrupts most weak connections between proteins.7
Formula:8
- 1% (watts/w) Nonidet P-40 (NP-40)
- 1% (watts/v) salt deoxycholate
- 0.1% (w/v) SDS
- 0.15 Michael NaCl
- 0.01 Michael sodium phosphate, pH 7.2
- 2 mM EDTA
- 50 mM salt fluoride (NaF)
- 0.2 mM new salt orthovanadate (Na3VO4.2H2O, it has phosphatase inhibitor functionality because it mimics phosphate9)
- 100 U/ml protease inhibitor, such as aprotinin
SDS (sodium dodecyl sulfate) lysis bufferedit
SDS is ionic denaturing detergent. Scorching SDS buffer is definitely often used when the protein require to become completely solubilized and denatured.
Formula:8
- 0.5% (watts/v) SDS
- 0.05 Meters Tris⋅Cl
- Adjust pH to 8.0
- Add 1 mM clean dithiothreitol (DTT)
ACK (Ammonium-Chloride-Potassium) lysing bufferedit
ACK is certainly used for lysis of reddish blood cells in biological examples where additional cells like as whitened blood tissues are usually of better attention.10
Formula:1112
- 150mM ammonium chloride
- 10mMeters potassium bicarbonate
- 0.1mMichael EDTA
- Adjust pH to 7.2-7.4
Lysis buffer in DNA and RNA researchedit
In research like DNA fingerprinting the lysis buffer can be used for DNA isolation. Dish soap can end up being used in a nip to break down the cell and nuclear membranes, allowing the DNA to be released. Other like lysis buffers consist of the proprietary Qiagen item Buffer P2.
Personal referencesedit
- ^Posch, Anton (2014-12-01). 'Sample planning suggestions for two-dimensional electrophoresis'.Records of Physiology and Biochemistry and biology.120(5): 192-197. doi:10.3109/13813455.2014.955031. ISSN1744-4160. PMID25211021.
- ^Peach, Mandy; Marsh, Noelle; Miskiewicz, EwaI.; MacPhee, DanielJ. (2015-01-01). Kurien, Biji T.; Scofield, Ur. Hal (eds.).Solubilization of Proteins: The Importance of Lysis Buffer Selection. Strategies in Molecular Chemistry and biology.1312. Springer New York. pp. 49-60. doi:10.1007/978-1-4939-2694-78. ISBN9781493926930. PMID26043989.
- ^Posch, Anton (2008).2D PAGE: Example Planning and Fractionation. Humana Press. p. 24. ISBN978-1-58829-722-8.
- ^acddMatters, EMBL - Workplace of Information and Open public. 'Protein Refinement - Extraction and Clarification - Selection of lysis buffer and additives - EMBL'.www.embl.de. Gathered2016-03-16.
- ^awLinke, Dirk (2009-01-01). Deutscher, Richard Ur. Burgess and Murray P. (ed.).Part 34 Detergents: An Review.Strategies in Enzymology. Guide to Protein Refinement, 2nd Copy.463. pp. 603-617. doi:10.1016/h0076-6879(09)63034-2. ISBN9780123745361. PMID19892194.
- ^'Detergents for Cell Lysis and Protein Extraction'.www.thermofisher.com. Gathered2016-03-16.
- ^atdJi, Hong (2010-08-01). 'Lysis of Cultured Cells for Immunoprecipitation'.Chilly Spring Harbor Methods.2010(8): pdb.prot5466. doi:10.1101/pdb.prot5466. ISSN1940-3402. PMID20679375.
- ^abchemicalSefton, Bartholomew Meters. (2001-01-01). 'Labels Cultured Cells with32Piand Preparing Cell Lysates for Immunoprecipitation'.Labeling Cultured Cells with 32Pi and Preparing Mobile Lysates for Immunoprecipitation.Present Methods in Molecular Biology. Section 18. Bob Wiley amp; Sons, Inc. pp. Device 18.2. doi:10.1002/0471142727.mb1802s40. ISBN9780471142720. PMID18265167.
- ^'Trial preparation for western blot Abcam'.www.abcam.com. Retrieved2016-03-16.
- ^https://www.thermofisher.com/order/catalog/product/A1049201
- ^'ACK Lysis Buffer'.Chilly Spring Have Methods.2014(11): pdb.rec083295. 2014. doi:10.1101/pdb.rec083295.
- ^'A10492 - ACK Lysing Barrier - US'.
Retrieved from 'https://en.wikipedia.org/watts/index.php?title=Lysisbufferamp;oldid=884606251'
